So I am only about 60 years late. I finally read Silent Spring, by Rachel Carson, and since I worked for decades in the agricultural chemicals industry, I have some thoughts about this book and all that it has inspired. What I find amazing about Carson’s work is how applicable it is to the world of today.
Rachel Carson wrote about the effects of the first generation of organic herbicides and pesticides. Those molecules were brute-force bludgeons against insects and weeds, with little discrimination against target species and collateral damage. Her description of the effects of indiscriminate spraying, coupled with the effects of resistance building in the insect populations, is just as valid today as it was when the book was written. And the praise she had for integrated pest management was also well ahead of its time (or maybe we are just now realizing how right it was).
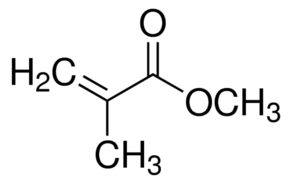
I chose to get involved in the agricultural chemicals arena. I accepted a transfer within my company, and one of the reasons was that the new generation of herbicides was manufactured at my new plant in West Virginia. It took a few years, but I was finally employed by the ag side of the plant. At that time our main herbicide was a truly specific offering, one that dealt with weeds but did not spread beyond where it was applied. It was something that fulfilled Rachel Carson’s dream, a chemical solution which did not cause collateral damage. Unfortunately, this was the time when Monsanto began to offer their solution of RoundUp Ready® products. These products offered the farmer a one-stop service, where they could spray a field with herbicide, knowing it would not bother the seedlings planted there which had been genetically modified for herbicide resistance.
We very quickly lost market share, and our good offering which I was proud of supporting, soon became yesterday’s news. We ended up licensing the technology for this genetically modified solution ourselves, and this allowed us to recapture a bit of market share though reducing our profits due to the licensing costs. But guess what? Farmers were supposed to vary their herbicides every couple of years to help prevent weeds from gaining resistance to the herbicide. The problem was that Monsanto offered such an easy solution for the farmers, what with its opportunity for no-till agriculture, very few farmers rotated herbicides. They tended to use the same one year after year.
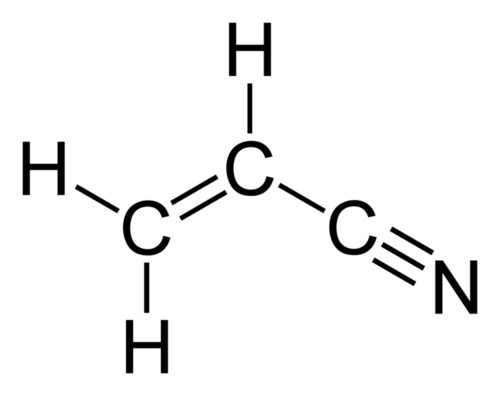
Guess what happened? Weeds began to gain resistance. So now you had fields with certain intransigent weeds peeking up through the intended crops, and the agricultural chemical companies sought a solution. Even though we still offered our environmentally friendly herbicides, the lure of no-till agriculture was now thoroughly embedded in the minds of farmers. So the answer developed was to add resistance to a second chemical in the seeds of crops. Monsanto / Bayer came up with an offering where their plants were resistant to RoundUp® and Dicamba, and their chemical offering was a blend of those two chemicals. Unfortunately, Dicamba would evaporate, especially in the warmth of the southern US, and its effects were felt far from the application site, causing uncontrolled damage. And, again, if farmers use this product exclusively, weeds will once again grow resistant to both chemicals. This will probably result in yet another chemical being added to the mix to aid the farmers in their attempt to eliminate tilling while still resulting in high crop yields.
I would have hoped that my company would have been more responsible, and come up with a solution requiring little additional chemical application. But no, my company’s preferred solution was to genetically modify the seed to become resistant to RoundUp® and one of the first generation of chlorinated hydrocarbons, 2,4 – D (2,4 – Dichlorophenoxyacetic acid). So the chemical arms race continued to run amuck, with the original goal of reduced chemical application long forgotten. I retired before this new product could be marketed, but I definitely did not like the direction we were heading towards.
The chemical race continues on insecticides as well. The first generation of broad-spectrum, chlorinated hydrocarbons, or the organophosphorus insecticides, were replaced by biodegradable compounds aimed at disrupting the life cycles of the insect targets. But even in the newer age of chemical warfare against insects, unintended consequences keep on popping up. The class of insecticides known as neonicotinoids has achieved broad use. Unfortunately, the effects on pollinators, both domestic honeybees, and wild bees, was much greater than expected. In addition, insects in general have been reduced, with unknown impact still to come from those portions of the ecosystem which depend upon insects for their food. Rachel Carson’s Silent Spring may yet come about again, due to birds starving and being unable to raise new generations of young.
The dream of integrated pest management Rachel Carson espoused has yet to come to pass. Speaking as one who was greatly invested in the business, as long as there is profit to be had from chemical application, companies will prefer to go after that profit instead of solving the real problems facing society. We still have a long way to go before we come up with ways to co-exist with the natural world instead of trying to compete and conquer those species we consider as our enemies.
A personal note here – Rachel Carson received her undergraduate degrees at the Pennsylvania College for Women. This institution changed its name over the years to Chatham College. It is there where my wife received her bachelor’s degree with a double-major in music and English. She is proud of her college’s famous graduate. What’s more, it is apparent that the city of Pittsburgh, home to this educational institution, is also proud since one of the bridges in downtown across the Alleghany River is named the Rachel Carson Bridge. In Pittsburgh there are three bridges connecting the North Side to downtown. Those bridges are the Rachel Carson Bridge, the Andy Warhol Bridge, and the Roberto Clemente Bridge. Truly an iconic mix of honorees reflecting on the eclectic mix of people associated with the city of Pittsburgh.