The laughs keep coming. Every day more and more cringe worthy statements come out of Republican mouths. The latest one is Trump’s excoriation of McConnell – a brief mention of his recent health scare, followed by scorn heaped upon him for supposedly signing on to the Green New Deal through supporting the infrastructure plan. See, what these “Publicans want you to know is that good ole American energy can only consist of things you burn. Coal, and natural gas, and oil, represent America’s birthright. Any suggestion that they cause harm goes against what all true American’s want. Thus the antipathy towards all forms of renewable energy – it’s not worthwhile to try to convert solar or wind to electricity because these sources don’t work 24/7.
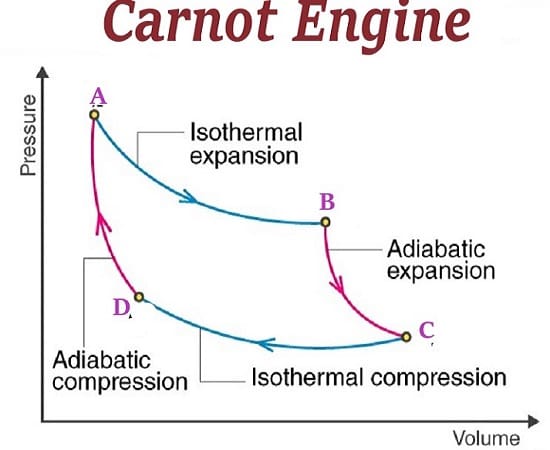
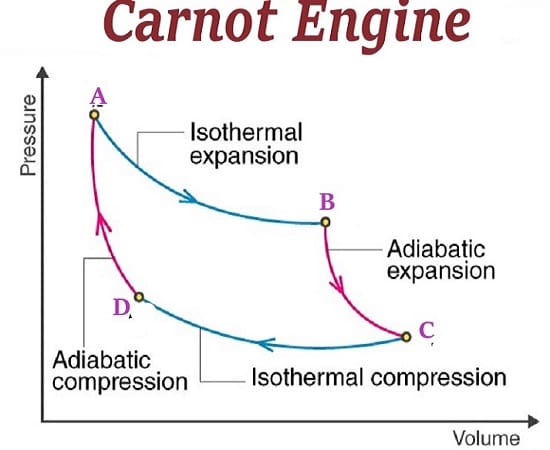
It is obvious that none of these fake patriots who support fossil fuels ever took a course in thermodynamics. There they would learn about the inherent inefficiencies in the Carnot cycle. Energy can only be extracted from the difference between two thermodynamic states. In general, this is high pressure steam, being reduced to a lower pressure after doing work turning a turbine inside of a magnetic field. This is how we generate electricity in power okants. Any fossil fuel combustion process uses this. Nuclear energy also uses steam cycles as well. But what these science illiterates either ignore, or are ignorant of, is that less than 50% of the available energy from a steam turbine is converted into electricity. That means that only ½ of the energy created from combustion actually goes to creating a form of energy useful to humanity. All the rest of the energy goes to die in thermodynamic hell as what is euphemistically called waste energy. You may be able to capture bits and pieces of this waste energy, through extraordinary means, but the fact remains that a large portion of the energy we release by burning goes into heating the environment. By the way, that is only a small amount of heat compared to what the greenhouse effect can capture.
The two main sources of energy available from renewable sources are solar energy, and wind energy. Both of these sources have conversion issues. For solar energy, the direct current must be changed to alternating current, and stepped up in voltage. Similarly, the output from windmills must be changed into a useful form for transmission along electrical lines. But guess what? The same conversion issues exist with fossil fuel sources. Steam turbines electrical output must be converted to high voltages so it can be conveyed to the ultimate consumer. What can be said about renewable sources of energy is that they do not have the inefficiencies of the Carnot cycle to steal from their useful output.

Yes, but only in fossil fuel plants and internal combustion engines can we take advantage of the high energy density available in our fuels. We would be wimpy if we tried to use only electricity to fuel our God-given right to mobility. We will never surrender to those who say there are limits to what we can and cannot do. Damn socialists!
You hear those statements coming from committed conservatives, who wish the status quo to remain in place. By God, we became great by using what was available in this nation, and no one can tell this 4% of the world’s population that we cannot use 15% of the world’s energy forever and forever. We are exceptional after all, and no one can steal our birthright.
Well, I hate to break it to the MAGA crowd, but we are not the only ones on Earth. And there are more and more people who are demanding a say in what is happening to them. As human population has surged over the past 100 years, so too has the number of people living at low elevations. These are the people who are most affected by sea level rise. So if we ignore their plight, we worsen the refugee crisis. Then the kneejerk reaction is to put up walls along our borders to keep out invaders. Sorry, but it might be instructive to look at the causes of refugee migration. By our insistence upon continuing to release ever increasing amounts of carbon dioxide, we threaten to inexorably turn our planet into a hell hole for many of the people we live with on Earth. And in case you haven’t noticed, there ain’t much hope for finding another world we can mess up.
During my lifetime, it is amazing how we have expanded our abilities to learn about other planetary systems. I see many stories about new planets being found in other solar systems, but nowhere have I seen a report on a habitable planet other than the one we live on. Besides, the planets we are looking at are many light years away. Do you have any idea how far a light year is? Do you have any idea how much energy would be needed to launch a sizable population to a new planet? And how many generations of humanity that would take? No, I am quite certain that those who control the money in politics are solely concerned with their own status as the lucky few who have “made it” in the world. By seeing clearly how far they potentially have to fall, they insist on using their money as political speech, writing multi-million dollar checks to political action committees whose intent is continuing the conflagration of fossil fuels. It does not matter that the projections of climate warming from increasing greenhouse gas concentrations are coming true on a daily basis. They find the one scientist who does not agree with the vast majority, and exclaim “Oh, it’s not a universal consensus. We can continue to claim that the science is not settled.”
So what will it take to convince these science deniers that potentially irreversible changes are occurring? And that they will not like the results? My guess is that their opinions will change only when heat domes descend upon their vacation homes, causing fuel drying and leading to catastrophic fires such as we’ve already seen in the west, and along the Mediterranean. After all, if these pampered people cannot fly their private jet into Aspen because it is on fire, they may begin to feel the effects of global warming. In which case my prediction is they will wonder why no one has taken action before it got this bad.